Configurable Business Documents
Configurable business documents (CBD) are special Electronic reporting (ER) configurations, which can be used instead of Print management SSRS reports in the posting and printing process of common business documents such as Sales invoice and Purchase order. Instead of SSRS print destinations, ER destinations are used.
ER configurations delivered by Microsoft for a single CBD consist of an ER Data model (data source schema), an ER Model mapping (mapping between the data source schema and the data) and ER Formats (Excel/Word templates including mapping between the data source schema and the template). CBD ER Model mappings map the data from SSRS report data sources, consisting either of tables populated by DP (Data Provider) classes or queries, to CBD ER Data models.
Docentric enables use of CBD ER Data models and Model mappings as report data sources. This way end-users and functional consultants can add additional data to reports by using Electronic Reporting Designer. Report templates are still designed with Docentric Designer and distributed via improved Docentric print destinations.
Learn more on SSRS, CBD and Docentric execution pipeline >>
Download and see the benefits >>
Enable CBD data sources
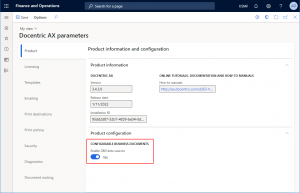
You can enable Configurable business documents ER Data models to be used as report data sources in Docentric AX parameters > Product > Product configuration.
Once you turn on the Enable CBD data sources option, the following menu items and form controls become visible in Docentric report setup:
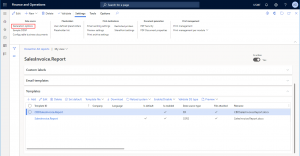
- The Data source > Configurable business documents menu item, which opens the Configurable business documents settings
- The Configurable business documents settings form, which enables configuration of ER Data model and ER Model mapping to be used with the report.
- The Templates > Validate DS type menu item, which for the selected templates, validate the Data source type field and update it to match the information from the template content.
- The Data source type field in the Templates grid, which indicates whether the template is SSRS- or ER-based. Data source type is set when reloading a System template or attaching the template file.
- The Data source generation options form: the Which DDSP to generate combo box, where you can select whether both SSRS and CBD DDSPs should be created or only the single DDSP that matches the template’s data source type.
See also
Setup and Use of CBD Data Sources >>
Docentric CBD Replicas >>
Comparison: SRS, CBD and Docentric execution pipeline >>
Webinar: Configurable Business Documents >>
Improved Configurable Business Documents >>